Do Calories Count? 1st Law of Thermo (Part 1)
April 26, 2011
This is a two-parter. Part 2 is here.
Calories are a contentious subject in the fitness and nutrition community, especially around these parts. The whole foods corner stresses that food quality is more important than quantity, and the low-carbers place the emphasis on hormonal balance; both of these stances can be extorted with little effort into the definitive “calories don’t count.â€
I must say, this calorie apathy is extremely tempting to buy into, but I can’t quite get myself to embrace it completely. On one hand, I know our hunter/gatherer ancestors’ dinner didn’t make a stop on the scale between beast and belly, and they did just fine. There absolutely must have been times of excess (I’d venture to guess some lived their whole lives in times of proverbial “feastâ€), so I don’t buy the claim that they stayed healthy due to lack of food availability. This is why I believe that staying healthy and lean should be relatively effortless – and certainly should not require scales or measuring cups.
On the other hand, it seems likely that there was a fluctuating food supply for some length of our evolution, so it stands to reason that the ability to store energy for later would have been selected for at some point. I just can’t imagine that a hunter letting extra calories slip away would have as good a chance of reproducing as his pack-rat neighbor. This is why I believe that one will store fat if they eat in tremendous excess for a significant period of time.
So here I sit (on the fence) thoroughly convinced that calories both count and don’t. It appears that evolution has provided the question and a few reasonable answers, but no concrete solution. Enter science.
Let’s just conduct some sort of study. You say we’ve tried that and haven’t gotten much of anywhere? But Taubes built such a convincing case with that smattering of research from when my parents were children. Oh, some research has been conducted in the last 30 years? I see.
Admittedly, I’m being a bit facetious here. The point I’m hoping to get across is that if you look at everything science has done on the subject you won’t come away with any clearer a picture than we painted above. Which is disappointing, but understandable – this stuff is just plain complicated.
The inevitable destination of discourse on this specific subject is the first law of thermodynamics. I must say, as someone who has had to sit through countless quarters of thermodynamics lectures, I often take offense to the treatment the first law receives. Without further ado, the first law of thermodynamics (click to enlarge):
In words: the rate of energy change within the control volume is equal to the rate of heat gained minus the rate of work performed plus the sum of the energy in incoming mass minus the sum of energy in the outgoing mass.
If we use the human body as the control volume, we seem to have arrived at the standard calories in, calories out conclusion. The Vi, zi, Ve, and ze terms can all be set to zero in this case, because we can assume that the kinetic and potential energy of food is not a factor here (certainly accurate). This gives us a simplified form of the first law:
We’re making progress. It seems that so long as we account for heat entering and leaving, work being performed by the body, and calories coming in and going out, we’ll have an accurate model of the human body. Unfortunately, as I discussed in this post, it’s pretty darn hard to get accurate values for these dynamic parameters. For the sake of this conversation, we’ll assume that we can.
[For those too lazy to link over, the conclusion is summed nicely by this analogy: The energy out of the body is a knife, and the “calories consumed†is a marble we’re dropping near the knife. Since it’s hard to accurately account for the position of the knife and the marble, you can only be sure that the marble will land on the desired side of the knife if you drop it far away (large excess or restriction).]
Even if we can accurately measure all this stuff, there’s still the issue of how calories are used. For example, if food enters the body in the form of a micronutrient we’re deficient in, those calories will be used for something other than energy. This is the first fundamental flaw in using the first law of thermo for diet: we have (practically) no way of knowing whether incoming calories will be used for energy, meaning that we don’t know whether they should be included in the “mihi†term.
It doesn’t really matter that this application of the first law is flawed anyway, because we aren’t looking where we care to find answers (an anecdote about a drunk and a streetlight comes to mind). We already know that incoming food can be burned, stored, or used as substrate for constructing or repairing cells. Let’s just look at storage. If calories get stored in our bodies, it makes a huge difference to us where. If my body is in storage mode after a big meal and puts a bunch of calories away in muscle, I’m a happy camper. If those calories go to fat cells, I’m less happy. The first law applied with the entire body as a control volume offers no resolution of the body’s subsystems, and therefore no context to the results of any model. So even if all accounting could be arranged and parameters calculated, we don’t care. It’s silly to discuss the merits of applying the first law to the human body as a whole, because even if it works, the results aren’t useful.
Unfortunately, we’re stuck looking at calorie intake and energy expenditure because it is easiest to measure (just like the drunk looks under the streetlight even though his keys aren’t there).
Summing up so far: Applying the first law to the whole body requires near-impossible accounting accuracy, but it doesn’t matter anyway because no one has the goal of reducing the energy storage of their body – they are interested in losing fat or gaining muscle.
In the second installment of this series, we’ll discuss how to apply the first law of thermodynamics to diet in a meaningful way.
Entry filed under: Basics, Calories, Food Choices. Tags: .
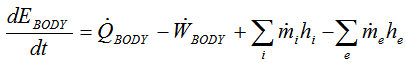



What’s Being Said